Atoms, Elements, and Compounds
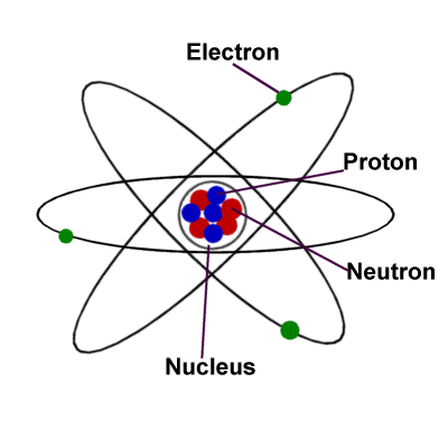
AtomsAtoms are the smallest unit of matter. They are the particles that make up everything that exists in the physical world. Atoms are composed of three particles, protons, neutrons, and electrons, that are found among two parts of an atom, the nucleus and electron orbitals.
Protons - these particles are positively charged and found in the nucleus. The number of protons that an atom has determines what element the atom makes. Neutrons - these particles have no charge and are found in the nucleus of an atom. Neutrons serve as a buffer between protons, preventing them for repelling each other out of the nucleus because they all have the same charge. The number of neutrons an atom contains determines what isotope of an element it creates. Electrons - Electrons are much smaller than protons and neutrons and carry a negative charge. They are found in the electron orbitals that surround the nucleus. If an atom gains or loses any electrons, the atom then becomes an ion, which means the atom carries either a positive or negative charge. |
Elements
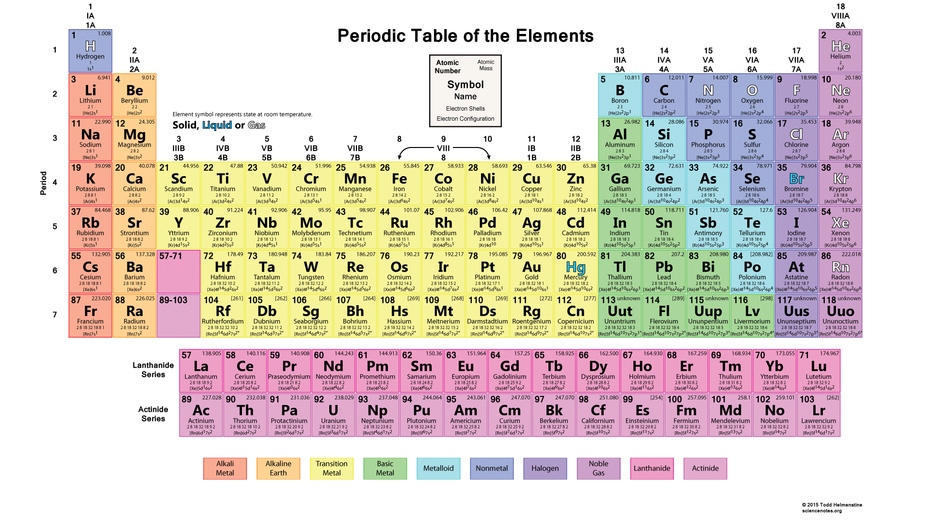
Elements are pure substances that cannot be separated into any other substances. There are over 100 different elements, with 92 of them that occur naturally. The elements have be organized and represented in a chart called the periodic table of elements. The vertical columns of the table are called groups and the horizontal rows are called periods. Elements of the same group will have similar chemical properties, for example, metals have a shiny surface and are malleable, which means that they can be shaped and formed.
Isotopes
When two or more atoms of the same elements have differing numbers of neutrons, those atoms are all isotopes of that element. We identify an isotope by placing a number after the element symbol or name. For example, one isotope of Carbon is Carbon-14 (C-14). The 14 represents the total number of particles in the nucleus of the carbon isotope (6 protons and 8 neutrons). When the number of neutrons in an atom is changed, it can affect the stability of the atom, causing the nucleus to break apart, or decay. When a nucleus decays, it gives off radiation. Because of this, most isotopes are radioactive.
Compounds
|
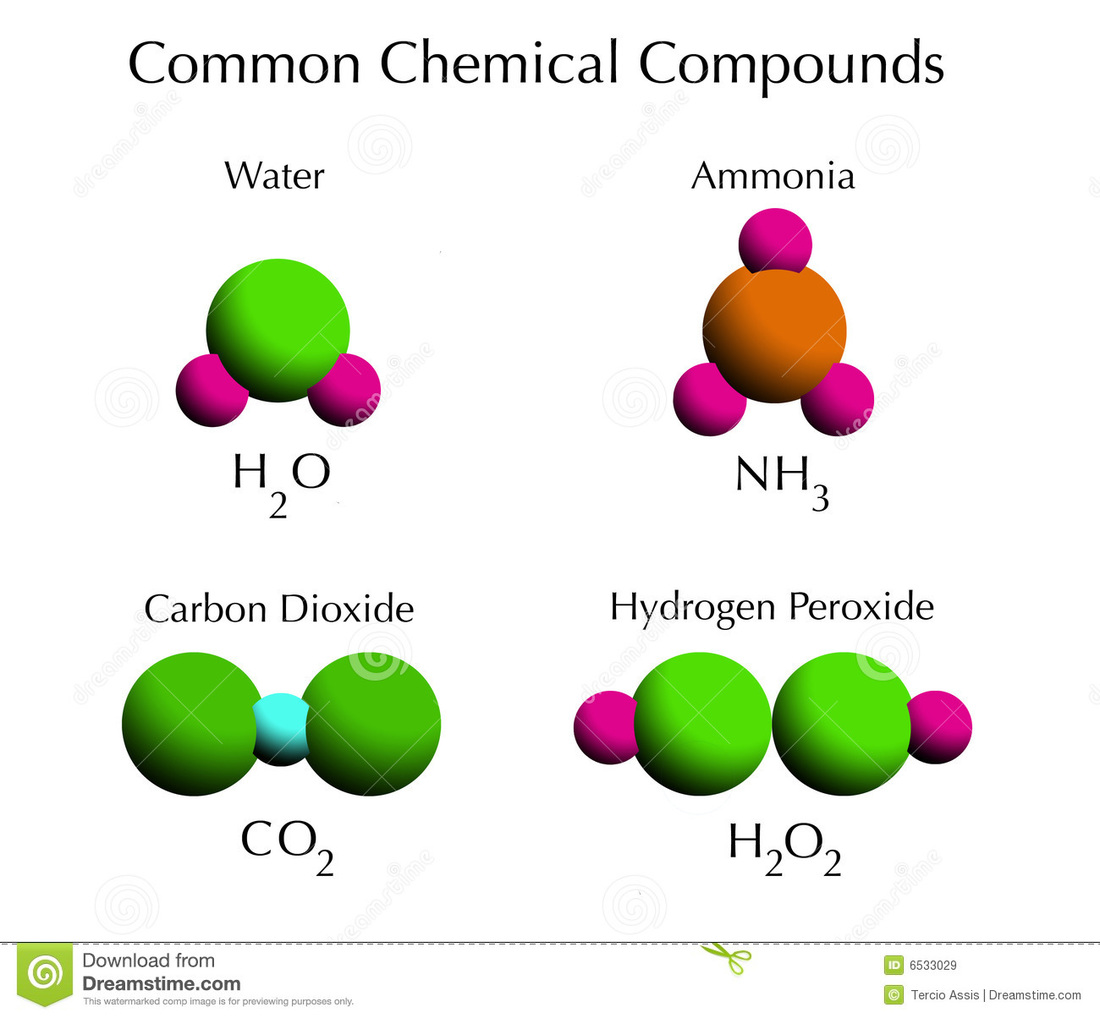
Compounds are pure substances that form when two or more elements combine. Common compounds are water, carbon dioxide, and ammonia. Compounds are held together by forces called chemical bonds. These chemical bonds are covalent bonds and ionic bonds.
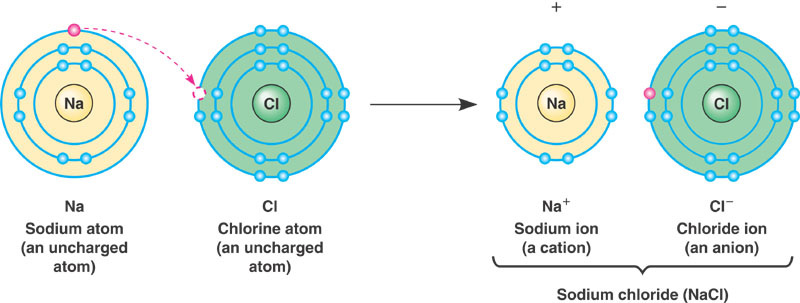
Ionic Bonds - these bonds occur when two ions, atoms that have either gained or lost electrons, have opposite charges and are held together by an attractive electrical force. An example of an ionic bond is when a sodium ion and a chlorine ion bond to create salt.
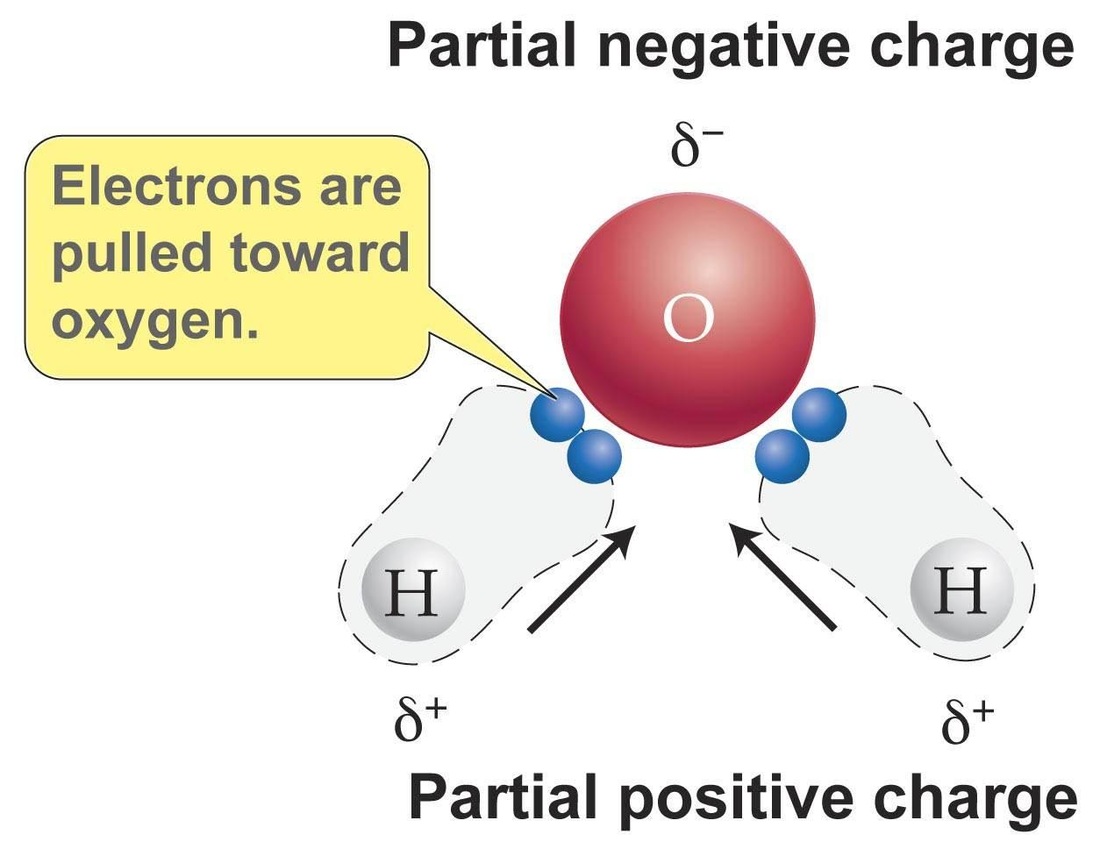
Covalent Bonds - these bonds occur when two atoms share one or more electrons to create a molecule. An example of a molecule held together by a covalent bond is water. |
Van der Waals Forces
|
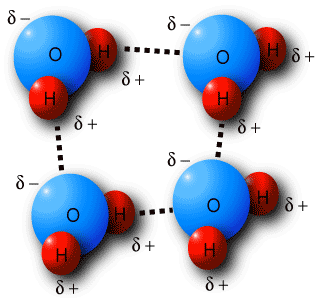
Because electrons move freely around an atom's nucleus in orbitals, when atoms bond covalently to create a molecule, electrons can "clump" together. A dutch physicist, Johannes van der Waals, discovered that these areas of slight charge on molecules create an attractive force that can hold molecules together. These van der Waals forces are very weak when compared to ionic or covalent bonds, but are the reason for why water molecules form droplets and how a gecko is able to walk on vertical surfaces.
|