Carbon: So Important,
There is a Whole Branch of Science Devoted to It
Carbon is so versatile that it's influence can be found in almost all life on Earth. Because of this all of organic chemistry is based around the study of carbon.
The Structure of Carbon:
|
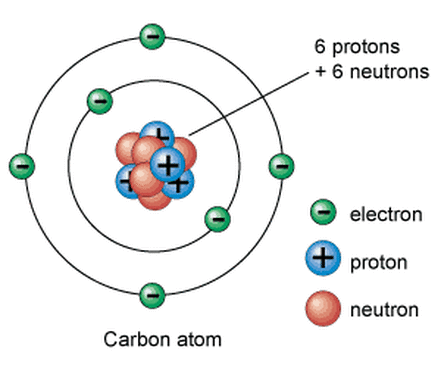
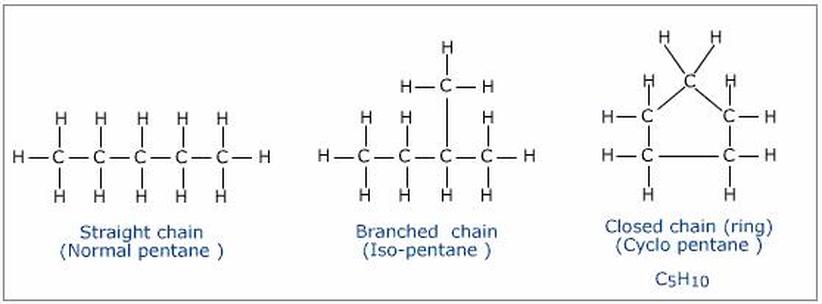
A carbon atom contains only four electrons in its outermost energy level. That outermost energy level can accommodate a total of eight electrons. Because of this, a single carbon atom can form covalent bonds with up to four other atoms. This is what allows carbon to be involved in so many different molecular structures that take on many different shapes, mainly straight chain molecules, branched molecules, and ring molecules.
|
Where all do we find carbon?
Carbon plays a key role in four main types of macromolecules: carbohydrates, lipids, proteins, and nucleic acids. Macromolecules are polymers, which are molecules that are long chains of smaller sub-units, called monomers, that are linked together. Many times, the monomers in a polymer can be identical, much like the links in a metal chain.
Carbohydrates:
|
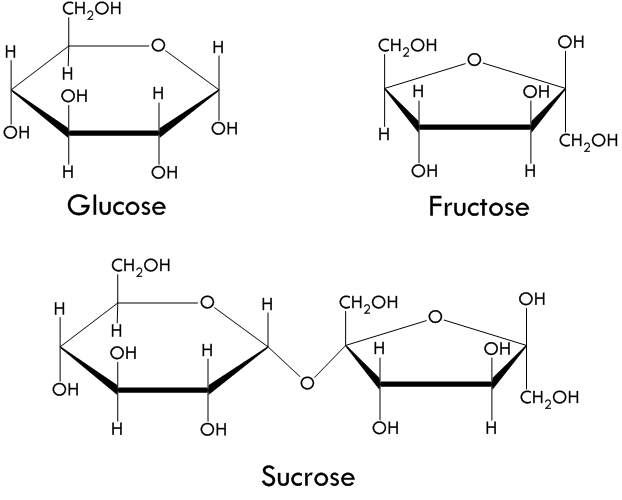
Carbohydrates - are compounds that are made up of carbon, hydrogen, and oxygen atoms in a ratio where there are two hydrogen atoms for every one carbon or oxygen atom. A general formula for Carbohydrates is (CH2O)n, where n is the number of CH2O units in the chain. When n is a value of 3 to 7, the carbohydrate is called a simple sugar, or monosaccharide. When the monosaccharides are linked together, they form complex sugars called disaccharides or polysacharides. Carbohydrates serve as sources of energy and provides structural support.
|
Lipids:
|
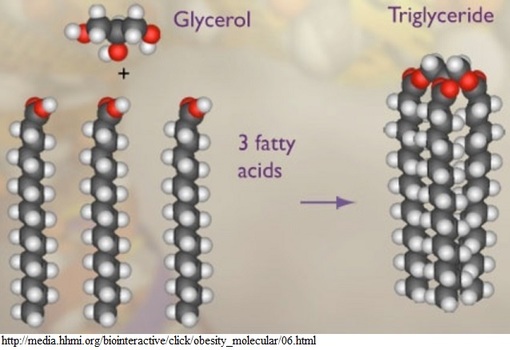
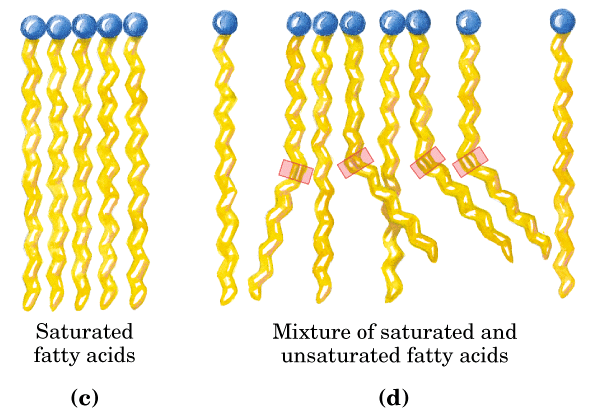
Lipids - Lipids are organic compounds that are made up of primarily carbon and hydrogen atoms and serve as a source of energy for most organisms. One category of lipids are triglycerides, which are made up of a combination of molecules called fatty acids and glycerols. The fatty acids are long chains of hydrogen and carbon atoms that are bonded together by single or double covalent bonds. The number of double bonds that occur in the fatty acid chains determine if the triglyceride is a saturated or unsaturated fat. Saturated fats are typically solid at room temperature, like butter, and unsaturated fats are typically liquid at room temperature, like oils.
Lipids also are found as phospholipids and steriods. Phospholipids make up the membranes of cells and steroids are what make up hormones. |